Below is a list of refereed articles based on our exciting research findings that we have published in various scientific journals up to this date. For easier access and a cleaner look, they have been grouped by year. Simply click on the title of the article to display the toggle abstract. The full PDF version is also available by clicking on the yellow button to jump to the publisher’s web site. If you do not have a subscription agreement with these electronic journals, please contact the Charette group to request a copy directly.
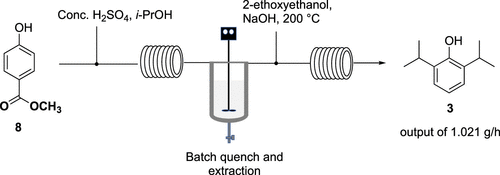
A multi-step process using continuous flow chemistry to produce propofol is described. A scale-up of a 5-stage process (two continuous flow chemical steps, two extractions using a semi-batch approach, and one purification) provided propofol in high purity. This process minimizes the number of impurities formed during the double Friedel–Crafts reaction allowing to run two continuous flow chemical steps sequentially. The use of simple, inexpensive, and readily available reagents affords a viable process for this widely employed active pharmaceutical ingredient.
In this communication, we describe the synthesis of novel hexahydroazepinone derivatives starting from two simple building blocks in presence of a readily available palladium catalyst. The reaction proceeds through a selective C(sp3)–H alkenylation/ring- opening process to obtain the seven-membered ring products in good to excellent yields on a wide variety of substrates under batch, microwave, and continuous flow conditions.
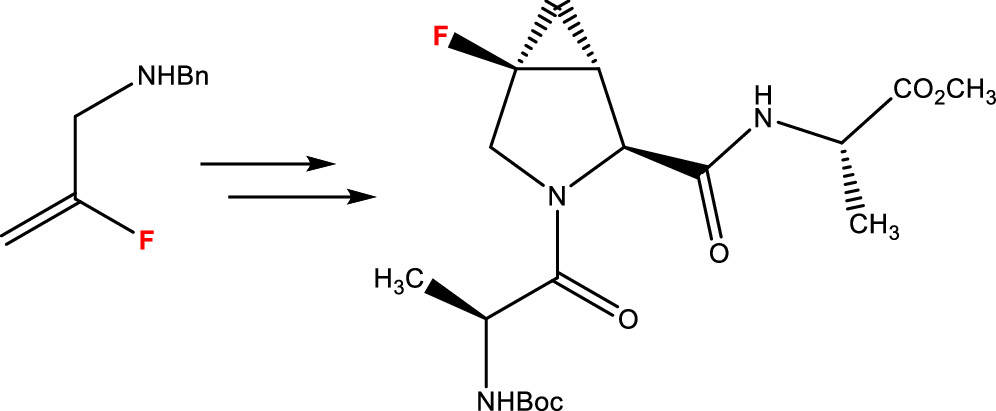
Over the years, numerous modifications to the structure of proline have been made in order to tune its effects on bioactive compounds. Notably, the introduction of a cyclopropane ring or a fluorine atom has produced interesting results. Herein, we describe the synthesis of a proline containing fluorocyclopropane. This modified amino acid was inserted into a tripeptide, whose conformation was studied by nuclear magnetic resonance and density functional theory calculations.
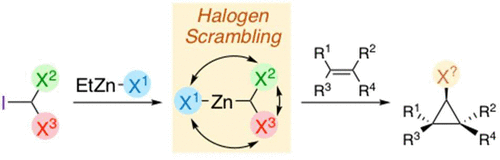
Dihalomethylmetal reagents are important species that can be used in halocyclopropanation reactions (metal = zinc) as well as in alkenyl halide synthesis. When trihalomethanes containing two or more different halogen atoms are used to prepare zinc carbenoids, several species can be formed through a halogen scrambling process. This affects which halocyclopropane, or haloalkene, is generated in the subsequent reaction. In the first part of the paper, we report an extensive characterization of zinc dihalocarbenoids by NMR to identify the major species formed when various di- and trihalomethane reagents are used as carbenoid precursors. In the second part of the paper, a direct application of this information enabled the development of a highly efficient chlorocyclopropanation reaction of allylic alcohols from inexpensive and accessible precursors.

An undergraduate teaching laboratory experiment involving a continuous flow, bleach-mediated oxidation of aldehydes under biphasic conditions was developed that allowed students to explore concepts of mixing or mass transport, solvent sustainability, biphasic reactions, phase transfer catalysis, and continuous flow chemistry.
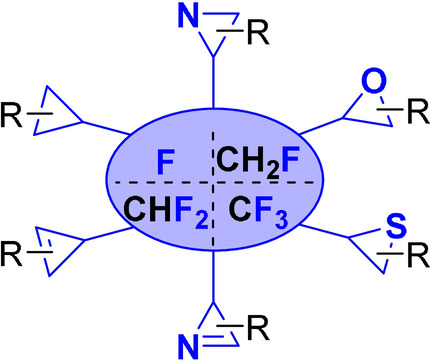
Better with three: Fluorinated three-membered rings are valuable compounds with many applications. In this Minireview, the synthesis of fluoro-, monofluoromethyl-, difluoromethyl-, and trifluoromethyl-substituted cyclopropanes, cyclopropenes, aziridines, azirines, epoxides, and thiiranes over the past decade are described.
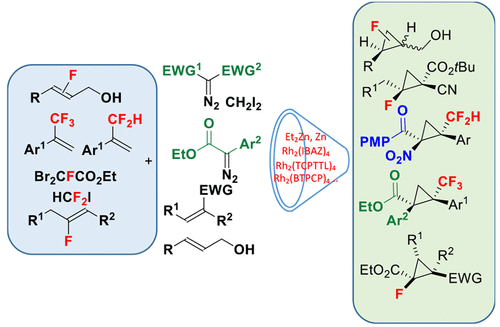
Fluorine-containing cyclopropanes are a subclass of cyclopropane derivatives that have generated considerable interest in medicinal chemistry for several decades. The replacement of a cyclopropane C–H or C–CH3 bond with fluorine or a fluorinated group (such as CF3 or CF2H) can lead sometimes to synergistic effects in terms of biological activity and improved metabolic profile of a cyclopropane containing bioactive compound. In this context, the preparation of fluoro-, difluoromethyl-, or trifluoromethyl-cyclopropane is particularly attractive and important but quite challenging considering the unique electronic properties that result from the incorporation of a fluorine atom into a substrate or a reagent. In the past decade, we have sought to develop new routes for the stereoselective synthesis of these building blocks using the most reliable cyclopropanation methods and convenient and readily available starting materials. The challenge that had to be undertaken was how we could use the unique properties of the fluorine atom to improve upon the efficiency of a given process rather than shutting it down. This could be overcome by defining new substrate/reagent reactivity guidelines and carefully selecting whether the fluorinated group was introduced on the electrophilic or nucleophilic partner for a given reaction. In this Account, we describe our contributions in this area that take advantage of diazo-derived rhodium carbenes, zinc carbenoids, ring closure processes, and biocatalytic methods to access these important potential drug subunits. Our initial investigation relied on the development of a Michael-initiated ring closure reaction using the Reformatsky enolate derived from readily available ethyl dibromofluoroacetate and α,β-unsaturated electrophiles. The reaction proceeded extremely well but with modest to good diastereoselectivities with ester acrylates. Further extension to various fluorinated nucleophiles such as oxazolidinone based and DABCO ylides led to similar selectivities.
In order to access enantioenriched fluorocyclopropanes, we then investigated the chiral dioxaborolane mediated zinc carbenoid based approaches using the fluoroiodomethylzinc carbenoid/allylic alcohol combination or the iodomethylzinc carbenoid/fluoroallylic alcohol combination. Quite surprisingly, both approaches were equally successful at providing the corresponding fluorocyclopropanes with excellent diastereo- and enantioselectivities.
To broaden the scope of fluorinated cyclopropane building blocks that could be prepared with good enantiocontrol, we then investigated the rhodium-catalyzed cyclopropanation of fluoro-, difluoromethyl-, and trifluoromethyl-substituted alkenes with acceptor–acceptor and donor–acceptor diazo reagents. Depending on the substrate/reagent combination, Hashimoto’s Rh2((S)-TCPTTL)4 or Davies’ Rh2((S)-BTPCP)4 catalyst proved be the most efficient catalysts providing the cyclopropane derivatives with the highest enantioselectivities.
More recently, a collaboration with Fasan’s group led to the use of engineered myoglobins to catalyze the reaction of ethyl diazoacetate and difluoromethyl-substituted alkenes. This biocatalyzed process led to high turnover number and high enantioselectivities.
Although our work has significantly increased the number of tools in the organic chemist’s toolbox, continuous efforts in this area would be beneficial to the development of diastereo- and enantioselective approaches to allow the preparation of any elusive isomers of these valuable chiral building blocks.

We report herein the development of two efficient synthetic routes for the preparation of a key fragment required for the synthesis of potent drug candidates of Hepatitis B virus. The ethyl 3-fluoro-1-H-pyrrole-2-carboxylate scaffold was synthesized from readily available starting materials in good overall yields. The scalability of one of the developed routes was demonstrated and afforded the desired target in good yield and excellent purity (99%).
Cyclopropanation reactions using zinc carbenoids are a powerful means to access cyclopropanes. Described herein is an enantioselective version of the reaction using zinc reagents and a chiral dioxaborolane ligand in the generation of fluoro- cyclopropanes. Readily available 2- and 3-fluoroallylic alcohols were efficiently cyclopropanated in high yields and excellent enantioselectivities. This method provides access to a variety of structurally diverse chiral fluorocyclopropanes that can be used as useful chiral building blocks.
An undergraduate teaching laboratory experiment involving a continuous flow, bleach-mediated oxidation of aldehydes under biphasic conditions was developed that allowed students to explore concepts of mixing or mass transport, solvent sustainability, biphasic reactions, phase transfer catalysis, and continuous flow chemistry.
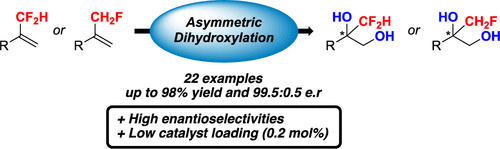
The catalytic asymmetric synthesis of α,α-difluoromethylated tertiary alcohols is described, using an asymmetric dihydroxylation reaction. This protocol using either the AD-mix-α or AD-mix-β allowed an easy access to these valuable fluorinated chiral building blocks, which have been obtained with excellent yields and er. In addition, the reaction was extended to the α-fluoromethylated analogues.
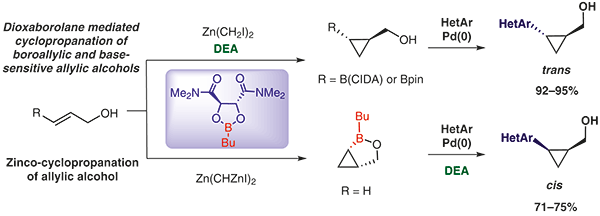
An enantioselective and non-oxidative methodology was developed to obtain enantioenriched cyclopropyl boronates using a diethanolamine-promoted selective decomplexation of dioxaborolane. The non-oxidative decomplexation of the dioxaborolane ligand from the cyclopropylmethoxide species formed in the dioxaborolane-mediated Simmons–Smith cyclopropanation reaction provided the enantioenriched CIDA-based (CIDA = N-cyclohexyliminodiacetic acid) borocyclopropane in 92% yield and 95.6:4.4 er. A robustness screen has shown diethanolamine to be compatible with esters, carbamates and N-heterocycles, providing a tool to access enantioenriched cyclopropanes carrying not only base-sensitive but oxidizable functional groups as well. Diethanolamine was found to be compatible with the modified zinco-cyclopropanation reaction of allyl alcohol to remove residual dioxaborolane from the corresponding cis-N-heterocycle cyclopropylmethanol, thereby leading to improved yields.
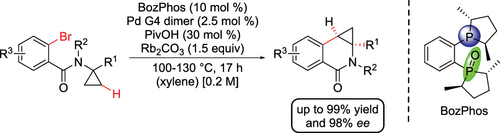
The bisphosphine monoxide (R,R)-BozPhos enables enantioselective C–H functionalization of cyclopropanes in a palladium-catalyzed cyclization. The synthesis of a broad spectrum of dihydroisoquinolones and dihydroquinolones in good yields and high enantiomeric excess was achieved through the use of this hemilabile ligand. Furthermore, the isolation of an intermediary palladium(II)-BozPhos complex after oxidative addition was successful and a second complex provided further insight into bond length and angles through a crystal structure.
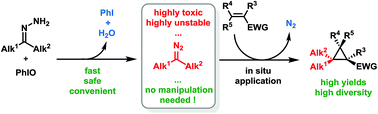
Electron-rich alkyl diazo compounds are powerful reagents in organic synthesis, but the risks associated with their toxicity and instability often limit their uses. Herein we describe an efficient, easy-to-handle and safe batch protocol for the in situ generation and cyclopropanation of these highly reactive non-stabilized diazoalkanes through the oxidation of free hydrazones using iodosylbenzene. Numerous substituted cyclopropanes have been synthesized using this methodology, including various gem-dimethylcyclopropanes of particular interest in medicinal chemistry.
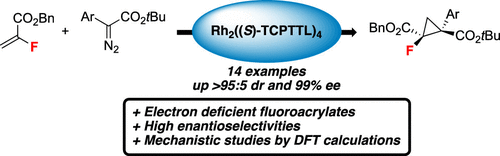
Herein, we report the catalytic asymmetric synthesis of functionalized fluorocyclopropanes from α-fluoroacrylates. The method using Rh2((S)-TCPTTL)4 allowed the difficult reaction of an in situ-generated electrophilic Rh-carbene with an electron-poor α-fluoroacrylate. The desired fluorocyclopropanes were obtained in good yields, excellent dr and ee. Finally, the mechanism of this transformation was studied by density functional theory (DFT) calculations to explain the particular reactivity of the donor–acceptor diazo compounds with electron-deficient α-fluoroacrylates.

The catalytic asymmetric synthesis of mono-fluoro-, -chloro- and -bromomethyl-1,2-diaryl cyclopropane ester is described. The reaction, using Rh2((S)-BTPCP)4 as a catalyst, allowed the formation of the desired cyclopropanes in good to excellent yields (up to 99%) and excellent diastereoselectivities (up to >20 : 1) and with a high level of enantioselectivities (up to 98% ee). Finally, the synthetic utility of the chiral cyclopropanes was also demonstrated.
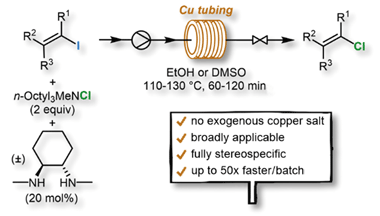
A simple continuous flow synthesis of alkenyl chlorides from the corresponding readily available alkenyl iodides in copper reactor tubing is described. A variety of alkenyl chlorides were obtained in good to excellent yields with full retention of the double bond geometry. The reaction time was reduced by a factor of 24–48 compared to the batch process.
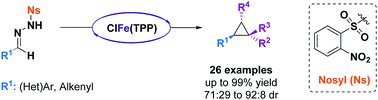
The modular synthesis of a variety of trans 1,2-disubstituted cyclopropanes in a safe and user-friendly one-pot iron-catalyzed cyclopropanation reaction is described. Easily synthesized N-nosylhydrazones are used as diazo precursors, allowing the in situ generation of electron-rich diazo compounds under mild reaction conditions and their direct participation in the cyclopropanation reaction.
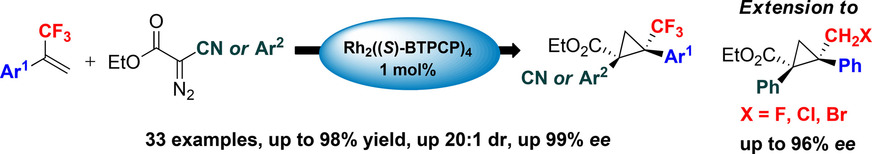
The catalytic enantioselective synthesis of CF3-cyclopropanes is described. The methodology afforded an access to highly functionalized trifluoromethyl cyclopropanes in high yields and high diastereo- and enantioselectivities. Extension to monohalomethyl cyclopropanes using the same chiral catalyst is reported.

Shine bright and efficiently: A user-friendly and metal-free UV-light mediated borocyclopropanation of styrenes using continuous flow technology has been developed. The use of xanthone, an organic photosensitizer, significantly shortened the reaction time, providing a highly efficient process. A broad range of borocyclopropanes can be obtained in good yields.
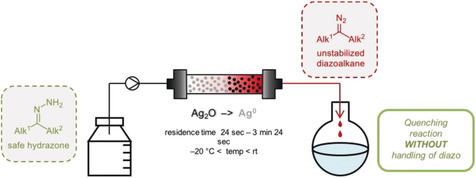
Red carpet treatment: Toxicity and safety issues still impede the common use of diazo reagents in academia and industry. Reported here is the safe and scalable production of the most sensitive and reactive diazo reagents, nonstabilized alkyl diazoalkanes. The ease of access to these reactive diazoalkanes allowed their use in esterification, cycloaddition, and Michael-induced ring-closure reactions.